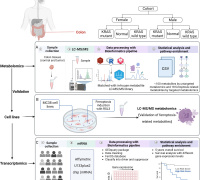
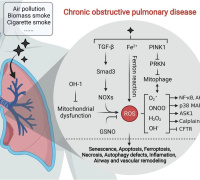
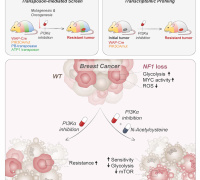
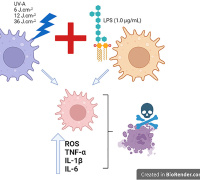
Join Redox Medicine 2024
 It is a great pleasure to announce the 26th International Conference on Redox Medicine 2024, which will be held on June 27-28, at Fondation Biermans-Lapôtre, Paris, France.
It is a great pleasure to announce the 26th International Conference on Redox Medicine 2024, which will be held on June 27-28, at Fondation Biermans-Lapôtre, Paris, France.
Speakers of Redox Medicine 2024
|
Small Molecule Electrophiles: Lessons from a Journey along the Redox Chemistry of Nitrogen Oxides to Drug Development Bruce Freeman, University of Pittsburgh, USA |
Keynote Day 2 Redox Interactome in Physiopathology Miria Ricchetti, Institut Pasteur, France |
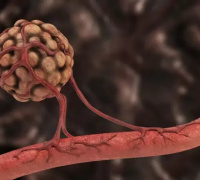
 Antioxidants Stimulate Blood Flow in Tumors Antioxidants Stimulate Blood Flow in Tumors Martin O. Bergo, Karolinska Institutet, Sweden |
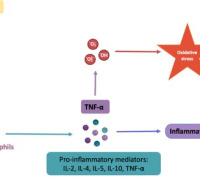
 Mechanism of the SuperInflammasome Complexes in Human Diseases and Redox Signaling Mechanism of the SuperInflammasome Complexes in Human Diseases and Redox SignalingMario D. Cordero, Universidad Pablo de Olavide, Spain |
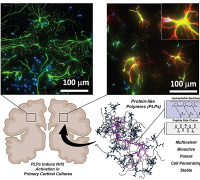
 Regulation of the Reactive Species Interactome by Prokineticin Antagonist in Cerebral Dementia Regulation of the Reactive Species Interactome by Prokineticin Antagonist in Cerebral DementiaChristel Marquette, Laboratoire Biosanté U1292 INSERM-CEA-UGA, Equipe MAB2, France |
 Epigenetic modulators link mitochondrial redox homeostasis to cardiac function in a sex-dependent manner Epigenetic modulators link mitochondrial redox homeostasis to cardiac function in a sex-dependent mannerZaher El Beck, Karolinska Institutet and Uppsala University, Sweden |
|
Tamara Tomin, TU Wien, Austria |
 The Reactive Species Interactome, Mitochondria, and the Gender-Brain Metabolism Nexus The Reactive Species Interactome, Mitochondria, and the Gender-Brain Metabolism NexusLaurent Chatre, ISTCT, CNRS, Université de Caen Normandie, France |
 NADH: The Redox Sensor in Aging-Related Disorders NADH: The Redox Sensor in Aging-Related DisordersRoberta Rizzo, Università degli Studi di Ferrara, Italy |
 Redox Implications of Supplementary Oxygen during Major Abdominal Surgery Redox Implications of Supplementary Oxygen during Major Abdominal SurgeryAndrew Cumpstey, University of Southampton, United Kingdom |
 New Insights into the Impact of Oxidative Stress in Hypertensive Diseases of Pregnancy New Insights into the Impact of Oxidative Stress in Hypertensive Diseases of PregnancyDaniel Vaiman, DR1 INSERM, Institut Cochin, France |
 Redox Medicine 2024: Biomarkers Redox Medicine 2024: BiomarkersCarole Nicco, President of Redox Medicine Society |
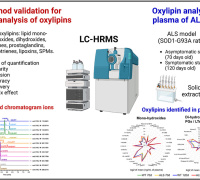
 Identifying Oxidized Lipids in Neurodegenerative Diseases Identifying Oxidized Lipids in Neurodegenerative DiseasesSayuri Miyamoto, University of São Paulo, Brazil |
 Exploring Predictors of HDL Oxidative Stress Functionality in Post-Covid Disease and Relation to Severity Exploring Predictors of HDL Oxidative Stress Functionality in Post-Covid Disease and Relation to SeverityJumana Saleh, Sultan Qaboos University, Oman |
 Redox, Radiotherapy and Biomarkers Redox, Radiotherapy and BiomarkersRadia Tamarat, Institut de Radioprotection et de Sûreté Nucléaire (IRSN), France |
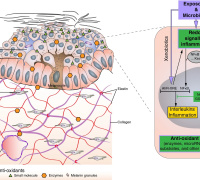
 Targeting the Keap1/Nrf2 Pathway for Treatment of Chronic Neurodegenerative Diseases Targeting the Keap1/Nrf2 Pathway for Treatment of Chronic Neurodegenerative DiseasesJeffrey A. Johnson, University of Wisconsin, USA |
 Antioxidant and Prooxidant Nanozymes: From Cellular Redox Regulation to Next-Generation Therapeutics Antioxidant and Prooxidant Nanozymes: From Cellular Redox Regulation to Next-Generation TherapeuticsGovindasamy Mugesh, Indian Institute of Science, India |
|
George M. Spyrou, The Cyprus Institute of Neurology & Genetics, Cyprus |
|
Duncan Emerton, Vice-President of Citeline, United Kingdom |
 How to Develop a Drug – From Preclinical to Clinical How to Develop a Drug – From Preclinical to ClinicalAntonio Vila-Coro, IQVIA, Spain |
 Artificial Intelligence in NeuroInflammation, Biomarkers and Redox Medicine Artificial Intelligence in NeuroInflammation, Biomarkers and Redox MedicineStéphane Mouchabac, CHU St Antoine DMU Neurosciences and Institut du Cerveau, Paris |
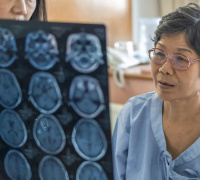
 Artificial Intelligence & Algorithms: Applications in Redox Medicine & Radiology Artificial Intelligence & Algorithms: Applications in Redox Medicine & RadiologyMaxime Barat, Université Paris Cité, Hôpital Cochin, France |
Redox Medicine 2024 Attendees
|
Citeline (Sponsor) |
Pusan National University |
Last updated on 12/04/2024